eSubmission Roadmap updated
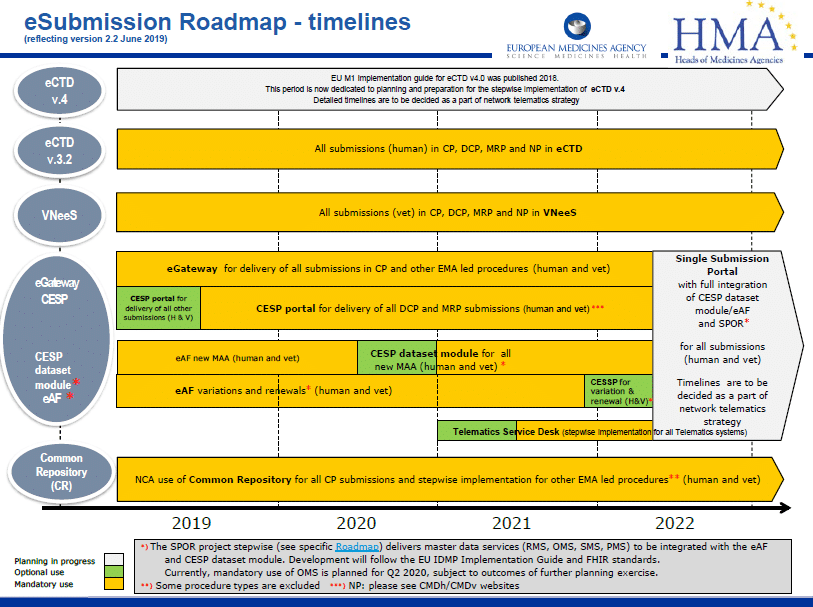
The European Union updated its eSubmission Roadmap in June 2019 to reflect changes in timelines.
What is it?
The eSubmission roadmap is a high level, strategic plan for business and technology changes within the EU. Its function is to align the plans and implementation timelines of target groups and stakeholders, including the EMA, National Competent Authorities (NCAs) and the pharmaceutical industry.
Full details on the eSubmission Roadmap are available on the EMA’s eSubmission website here.
What is happening?
Centralised Procedure(s) (CP)
It is mandatory since January 2010 that all submissions in the CP are made in the eCTD format
National Procedure(s) (MRP & DCP and national only procedures)
It is mandatory since July 2015 and January 2017 that all submissions for new MAAs using the DCP and MRP respectively are made in the eCTD format. For purely national procedures, eCTD has been mandatory for new MAA submissions since July 2018.
For all new submissions in MRP and DCP, the mandatory use of eCTD has been in force since January 2018, and for all new national submissions, since January 2019.
What does this mean?
This means that ALL submissions in the EU for human medicinal products now have to be made in eCTD format. Are you ready? If not, contact us to find out how we can help switch all your current MAs to eCTD and manage all your lifecycle needs. Ivowen has been eCTD compliant since 2009, and therefore has a wealth of experience to become your key partner in this vital step of your registration process.
If you need any clarification or support to manage the changeover to eCTD, Ivowen will gladly work with you to ensure a seamless and efficient transition to eCTD. Contact us for more information or to make an enquiry.
Written by Majella Ryan