Points to note on the eAF
General
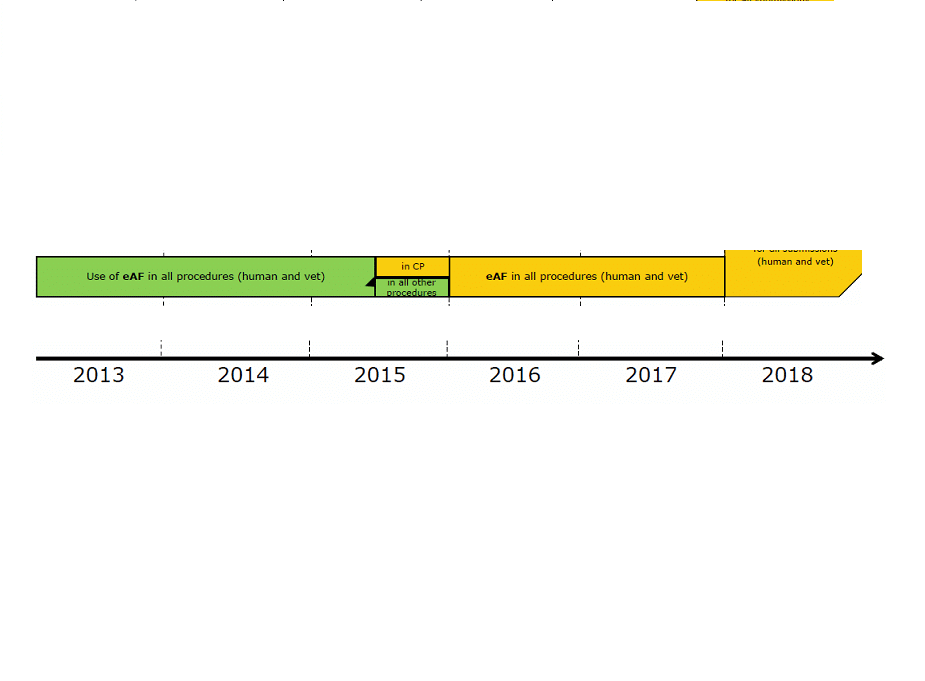
The eAF is mandatory for all procedures from 01/01/2016 (CP, MR, DC, National in eCTD/NeeS/Paper). However, Presubmission Meeting Requests, Article 61(3) & MAT forms remain the outside scope and can be submitted as paper or PDFs.
Please contact us if you require any assistance with any of the forms or any advice on any of the above procedures. For your convenience, the following is a summary of the important aspects of using the eAF.
- Always check the e-submission website for the latest version of the eAF (http://esubmission.ema.europa.eu/eaf/)
- Word versions will be removed from Notice to Applicants in January 2016
- After 11/01/2016 only version 1.19 will be acceptable for new procedures
- eAF should be printed for Paper submissions
- Version 1.20 is planned for April 2016 (unless hotfixes are required before then)
- It is important to note, and welcome news, that there is no need to update to a newer version of the eAF in the middle of a procedure
- See the guidelines (update planned in January 2016) and release notes (lists the changes made to newer versions) for further information
- In line with the proposed move towards a Single Submission Portal (SSP) in place of CESP and the EMA Gateway, there are plans to reformat the eAF into a data capture system that can be submitted directly through CESP. This is in a very preliminary stage.
- Technical queries should be sent to EMA Service Desk via IT service portal (login)
- Q&A documents should be consulted first
- Fast web view warning in the dossier validation report for the eAF can be ignored
- Procedural queries should be directed to the National Competent Authority (NCA) (and response sent to EMA Service Desk). Complicated queries should be sent to both parties.
- Webinars are available for the NCAs (to try to reduce the requests for MS specific national requirements)
Using the eAF
- Technical Validation of the form (internal):
- Once signature is added and form validated, it is now locked. Locked forms cannot be amended. Therefore, the signature should be the last thing added to the form.
- Always keep a copy of the unlocked form so that amendments can be made (e.g. during preparation or for requests from NCA for updated forms during a procedure)
- Do not use bookmarks as these may cause invalidation issues
- Annexes to the form should be filed separately in module 1.2 (do not use the PDF function to insert them into the eAF)
- Form should be named; cc-form-eaf-var
- Annex should be named; cc-form-annex-var (e.g. cc-form-5-19 or cc-form-proofpayment)
- Electronic signature can be an image of a real signature (e.g. jpeg file – a scanned copy of wet signature. This however is not an electronic signature and is only used to close/lock the form) or can also be a line of text which states that signature is on file internally (e.g. “This form was authorised following company policies by Majella Ryan, Regulatory Affairs Manager of Ivowen with authorisation to sign. The signature is on file”)
- The eAF does not accommodate multiple signatures at present. A separate annex should be provided if multiple signatures are mandatory for a particular NCA. Multiple name sections will be incorporated into version 2.0 but no mention of whether multiple signatures will also be accommodated.
- The signature should be provided by the responsible MAH or can be provided by any authorised deputy
- Please also check national requirements for signatures.
- See Q&A guidance for further information
- Workaround solutions (e.g. Annex B for multiple MAH or Product names) should always be mentioned in your cover letter
- Some unforeseen variations (category z) are not adequately accounted for yet. Details of such changes should be outlined in the scope section of form and in the cover letter.
- Duplicate sections only if products differ with regard to API or Pharmaceutical Form
- Annex A or Annex B can be used for multiple MAH or Product names. Click on Annex A/B button in form and add the annex as a separate file in Module 1.2
- Detailed instructions on how to use workaround solutions is available in the eAF Q&A document and the eAF Technical Guidance documents – both are published on the eAF webpage – if you need more advice contact EMA Service Desk.
- No translations of the eAF are available, nor should they be requested.
- Drug substances can be entered from controlled vocabulary lists or free text, and these each have different EV codes.
- The focus should be on using substance, product, organisation and referential (SPOR)
- Request for New Terms:
- EUDRACT terminology used
- Use eAF Term Request form to request new terms
- Send it to mdms@ema.europa.eu only
- Response should be received within 5 working days
- Strikethrough text function is not available in eAF but text can be copied and pasted (with text struck through) from Word or Outlook
- In MRP/DCP one common application form is highly recommended, one per pharmaceutical form or strength for all member states in case of new MAA and one eAF for all involved products for all member states in case of variations and renewals.
Written by Majella Ryan